Thanks to the endeared Henrietta Lacks, a woman whose cervical cancer cells have majorly contributed to numerous scientific and medical breakthroughs and advancements, the medical community understands so much more about human health and how to treat a myriad of diseases. Although she passed in 1951, HeLa cells, as they are called, are still used as a fundamental component of advanced research. Henrietta’s life and contribution prove that black people have historically played and continue to play a vital role in the advancement of healthcare.
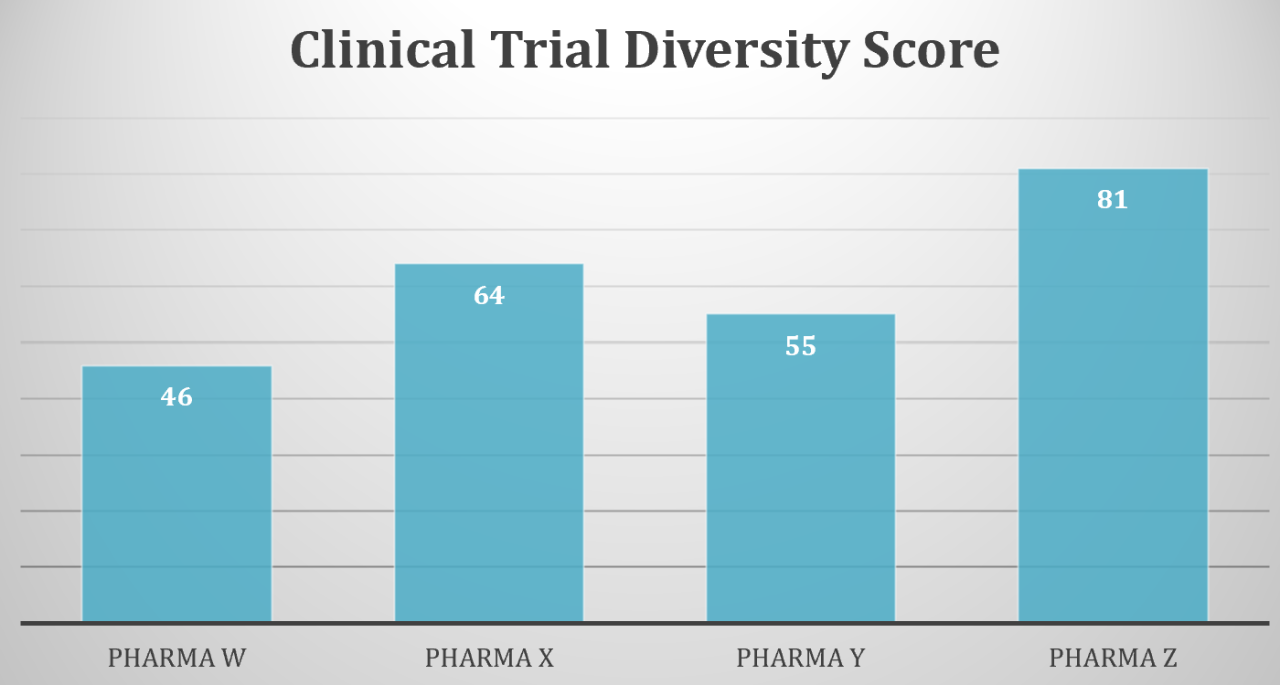
According to the FDA, “racial and ethnic minorities are underrepresented in clinical trials. This is a concern because people of different ages, races, and ethnicities may react differently to medical products.” In recent years, FDA Drug Trial Snapshots show that less than 10% of drug participants were black compared to around 80% for white participants. This massive contrast raises many concerns that Black/African Americans may not be getting the best type of treatment and drugs unique to their demographic.
Clinical trials determine safe and effective medical products, with specific protocols that can establish several factors involving screening, diagnosis, treatment, prevention, epidemiology, quality of life, and genetics. Because these trials are by and large performed on whites, there’s a gap in the real knowledge of how effective these products are for black people, how they can be improved, or whether or not they should even be given. This is important because it has been scientifically proven that certain diseases affect the black community more or in different ways than white people.
The main reason for the lack of representation of blacks participating in clinical trials is the majority of Black/African Americans’ distrust of the medical community. Efforts are being made to raise awareness of the need for the general medical community to address that distrust and until that is done there will likely be little progress. Black people simply need to see that white medical professionals understand why there is great hesitation and to address the issue vehemently.
Restoring trust can be done. It is the fundamental course of action before getting black people to even hear why their participation in clinical trials is so important. Yet, it can be done. When black people are convinced that their best interests are in the foreground, they will hear the gravity of why less than 10% for clinical trials, in general, is just not in their best interest. Simply put, where they are not seen, they cannot be heard.
To learn more about how Acclinate increases affective trust with communities of color so they are more likely to participate in clinical trials, start here.