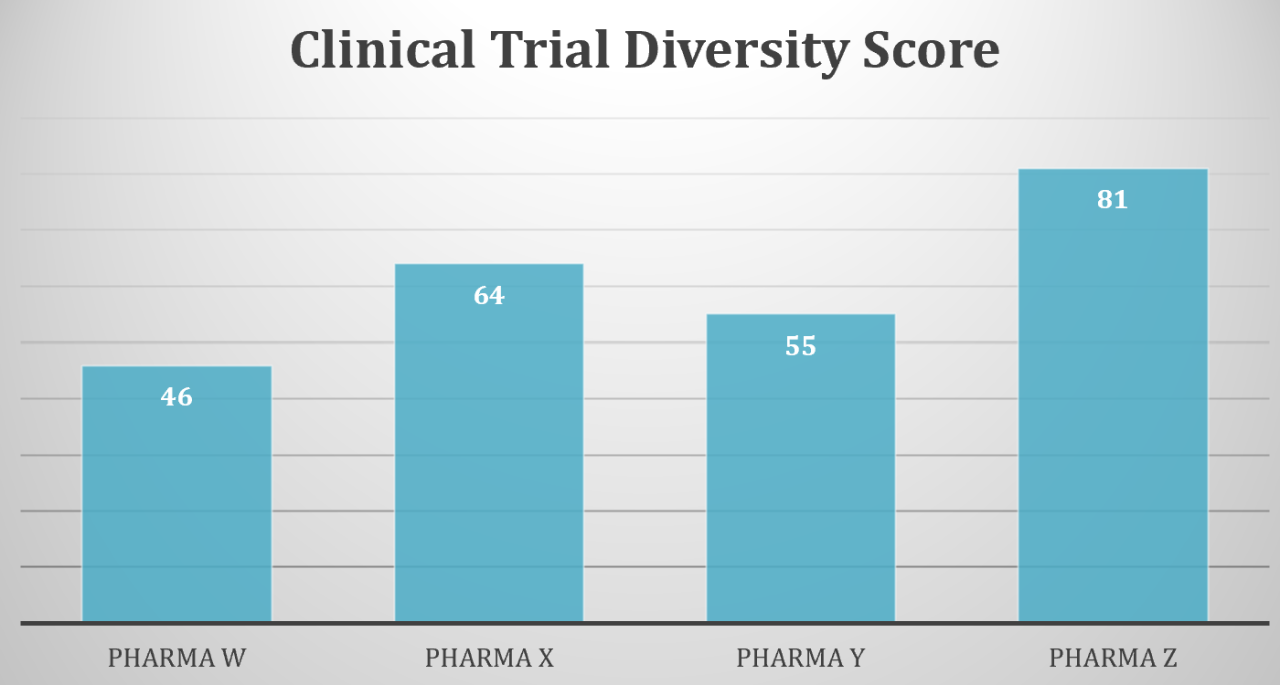
According to a recent study published in the journal Gastroenterology, Blacks and Hispanics remain noticeably absent from pancreatic cancer clinical trials. In fact, data showed that the actual inclusion of racial and ethnic minorities has stayed flat over 15 years. This research mirrors other studies revealing that Black and Hispanic races are consistently underrepresented in oncology trials compared with their burden of cancer incidence. And the problem of clinical trial diversity is not exclusive to oncology trials. For example, the American Diabetes Association recently highlighted large disparities in racial and ethnic representation among pivotal type 1 diabetes trials used to gain FDA approval, even more concerning given that rates of type 1 diabetes are increasing in underrepresented groups.
Pharmacogenomics informs us that people of different races and ethnicities may have different reactions to pharmaceuticals, so having a diverse population participate in trials helps ensure the safety and efficacy of new medications. Inclusive research also enables more patients to benefit from therapies that can improve their quality of life. However, as the two studies point out, the lack of racial and ethnic diversity in clinical trials is not new. For better or worse, COVID and the associated social justice movements have put a spotlight on long-standing health inequities, including clinical trial diversity. Fortunately, we have witnessed the FDA, pharmaceutical companies, CROs, and academic medical centers all moving the issue of clinical trial diversity toward the front of the priority line.
But why are clinical trial diversity initiatives still falling short of their expected goal even after all this attention and shifting of priorities?

New Approaches, Same Flaw
The guidance issued last year by the FDA, “Enhancing the Diversity of Clinical Trial Populations--Eligibility Criteria, Enrollment Practices, and Trial Designs,” provides recommendations for how sponsors can increase enrollment of underrepresented populations in their clinical trials, including steps to broaden eligibility criteria in clinical trials through inclusive trial practices, trial designs, and methodological approaches.
Several groups have focused on helping clinicians understand communication gaps that impede trial enrollment while helping patients gain insights into the value of clinical trials. Still, others are taunting the momentum of decentralized clinical trials (DCT) as the swift answer to clinical trial diversity. The rapid adoption of DCT due to the COVID-19 pandemic has revealed numerous benefits, including reducing barriers such as transportation and limited site access, accelerating clinical trial timelines, streamlining clinical operations, and reducing patient and investigator burden. However, the verdict is still out as to whether DCT is the answer for clinical trial diversity. It is likely that decentralized trials will help, but it will not solve the representation challenge in clinical trials.
A lapse in DCT trials is that they are less likely to be held in black communities where residents might be more likely and willing to participate. Even though DCT trials make a concerted effort to provide resources like transportation and increased access, another missing component may be the presence of nurses and administrators of color who understand the daily lifestyle of minority participants and who are better equipped to properly communicate with them.
Many of these approaches presume that the primary deterrent of clinical trial diversity centers on issues of protocol design, education, or access. These approaches underestimate the importance of affective trust when it comes to getting a potential participant to say “yes, I want to participate” through an informed consent process.
Not All Trust is Equal
Cognitive trust occurs when a person makes a conscious decision to trust-based upon the best knowledge he or she has. When interactions are based on cognitive trust, individuals choose to trust based on evidence of trustworthiness. Thus, cognitive trust tends to be high when repeated interactions allow parties to come to know, understand, and predict one another. Affective trust, on the other hand, develops when individuals emotionally invest in relationships, resulting in genuine concern for each other's wellbeing. As such, affective trust resides at an interpersonal level.
Here is an illustration of affective trust. You have a persistent cold. A quick WebMD search relays various courses of action recommended by a pulmonologist. You decide to call your grandmother who says, “just put some Vicks on your chest”. Because you know your grandmother has a genuine concern for you and your best interest, despite not being an MD, you lean toward the Vicks approach. Unfortunately, these are not isolated occurrences. People in communities of color handle medical situations as illustrated because of the inherent lack of trust in the medical community.
At the least, building cognitive trust has to become a priority in order to establish relationships between industry and minority communities. For most in those communities, both history and present interactions have further widened the chasm of trust.
So the question becomes: How do you repair that trust?

How Does this Apply to Clinical Trial Diversity?
As the adage goes, “People don't care how much you know, until they know how much you care.” The question that racial and ethnic minorities are implicitly wondering is whether, regardless of if they participate in a trial or not, researchers and practitioners will still care about their health and subsequently do what’s best for them. Communities of color are looking at pharmaceutical companies, CROs, and academic medical centers conducting clinical trials to ultimately answer this question. Even still, they don’t quite understand or trust them. To start to build, and eventually gain trust, the pharmaceutical industry can use innovation to address this fundamental flaw. Industry does not currently possess the innovation itself so it is incumbent that it builds relationships with innovators who have identified ways to build effective trust with communities of color.
What is the Solution to the Lack of Diversity in Clinical Trials?
Acclinate is the solution. The company serves as a bridge between communities and medical entities. Acclinate is able to help clinical trial sponsors save time, save money, and accelerate the time to market using a two-prong approach. First, #NOWINCLUDED is the growing network of minorities who are provided with education, story-telling, and ongoing conversation so that they are able to learn more about and be encouraged to participate in medical research. Secondly, the #NOWINCLUDED network data feeds into Acclinate’s HIPAA-compliant software platform, known as e-DICT™ (Enhanced Diversity in Clinical Trials). E-DICT uses predictive analytics and machine learning to identify the people who are most willing and likely to participate in clinical trials.
Essentially, #NOWINCLUDED does the front-end work of engaging and educating the community so they are empowered to participate. On the back end, E-DICT connects the industry with the people who want to be included as part of the solution.
To learn more about how Acclinate increases affective trust with communities of color so they are more likely to participate in clinical trials, request a demo.